- Nitrogen Electrons Valence Shell
- Nitrogen Valence Electrons Atom
- Nitrogen Valence Electrons Count
- Nitrogen Valence Electrons Number
- Nitrogen Valence Electrons
Hydrogen Cyanide is a colorless, flammable, and poisonous chemical liquid. Represented by the chemical formula, HCN is one of those molecules that has an interesting Lewis structure. This liquid is used in electroplating, mining, and as a precursor for several compounds.
The shapes of the five orbitals occupied in nitrogen. The two colours show the phase or sign of the wave function in each region. From left to right: 1s, 2s (cutaway to show internal structure), 2p x, 2p y, 2p z. A nitrogen atom has seven electrons. The valence electrons in nitrogen experience the charge of the nucleus through the slightly greater than 5+ shield of core electrons. The shielding experienced by any one of the valence electrons resulting from the core electrons is 1 nearly greater than 3. For the 2s electrons, the shielding from the other 2s electron is 2 4. Electronic configuration of Nitrogen is 2,5.So,the number of valence electrons in it are 5.So,the valency is 8-No. Of Valence electrons=8–5=3. So,the valency of Nitrogen is 3 and it exists in nature as a diatomic molecule (N2) making triple bond with another Nitrogen atom.
3; a nitrogen atom has 5 valence electrons and achieves and octet by gaining 3 electrons How many electrons must S gain to achieve a stable electron configuration 2; a sulfur atom has 6 valence electrons and achieves and octet by gaining 3 electrons. The rings of the electron cloud that hold electrons. The number of orbitals in 1s. Valence electrons vary. 1 valence electron. 2 valence electrons. 3 valence electrons. 4 valence electrons. 5 valence electrons.
And to further understand Hydrogen Cyanide’s physical properties, it is vital to know its Lewis structure and molecular geometry. Keep reading this post to find out its shape, polarity, and more. First, let us look at its Lewis dot structure and the valence electrons that participate in forming bonds.
HCN valence electrons
To draw the Lewis dot structure of any molecule, it is essential to know the total number of valence electrons in the structure. To know the valence electrons of HCN, let us go through the valence electrons of individual atoms in Hydrogen Cyanide.
This molecule is made up of three different atoms: Hydrogen, Carbon, and Nitrogen.
Hydrogen has one valence electron, and it only needs one more electron to complete its valence shell as it is an exception to the octet rule.
So Hydrogen has one valence electron.
Whereas Carbon has four valence electrons and Nitrogen has five valence electrons.
Total number of valence electrons in HCN= No. of valence electrons in Hydrogen + No. of valence electrons in Carbob+ No.of valence electrons in Nitrogen
= 1+4+5
= 10 valence electrons
Hence, Hydrogen Cyanide, HCN, has ten valence electrons.
HCN Lewis structure
Once you get the total number of valence electrons, you can make a Lewis dot structure of HCN. This structure helps in understanding the arrangement of valence electrons around the atoms in the molecule. It also aids with understanding the bonds formed in the molecule and the electrons not participating in any bond formation.
To start with making the Lewis Structure of HCN, we will first determine the central atom. And then place the remaining atoms in the structure.
As Carbon is the least electronegative atom in this molecule, it will take the central position. Place the Hydrogen and Nitrogen atoms on both terminal sides of the Carbon like this:
Once you have arranged the atoms, start placing the valence electrons around individual atoms. Like Hydrogen will have one electron, Carbon will have four electrons, and Nitrogen will have five electrons around its atom like this:
If you look at the structure closely, you will realize that Hydrogen can share one electron with the Carbon atom and become stable. So both Carbon and Hydrogen will share two electrons and form a single bond.
H-C N
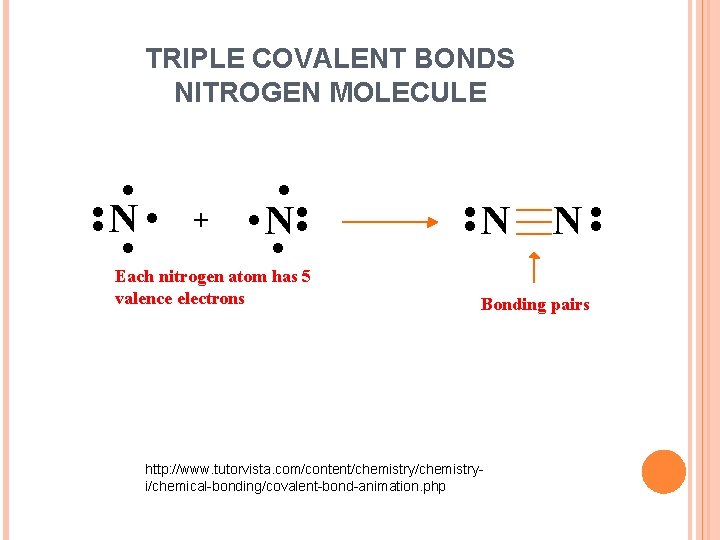
Now that we have completed the valence shell for Hydrogen let us do the same for the Carbon atom. The atom is left with only three valence electrons as it has shared one electron with Hydrogen. And so Carbon will share its remaining three electrons with Nitrogen to complete its octet, resulting in the formation of a triple bond between Carbon and Nitrogen.
Carbon has a complete octet by forming a single bond with Hydrogen and a triple bond with the Nitrogen atom. Similarly, Nitrogen has a complete octet as it only needed three electrons for completing the octet that it got by sharing the electrons with Carbon. Hydrogen has two electrons in its outer valence shell. The rest two electrons are nonbonding electrons.
HCN Molecular Geometry
The molecular Geometry of any given molecule helps understand its three-dimensional structure and the arrangement of atoms in a molecule, and its shape. Hydrogen Cyanide has geometry like AX2 molecule, where A is the central atom and X is the number of atoms bonded with the central atom.
As Carbon is bonded to two atoms, it follows the molecular geometry of AX2. And as per VSEPR theory, molecules covered under AX2 have a linear molecular geometry.
Hence Hydrogen Cyanide has linear molecular geometry.
HCN Bond Angles
Once we know the Lewis structure and Molecular Geometry of any molecule, it is easy to determine its bond angles and polarity. As this molecule has a linear molecular geometry, HCN has bond angles of 180 degrees.
HCN Shape
As both Hydrogen and Nitrogen are placed far from each other at bond angles of 180 degrees, it forms a linear shape.
HCN Polarity
HCN in a polar molecule, unlike the linear CO2. And here is why:
Carbon has an electronegativity of 2.5, Hydrogen’s electronegativity is 2.1, and Nitrogen has an electronegativity of 3.

Although Hydrogen is the least electronegative, it can never take a central position. And due to the difference in electronegativities between Carbon and Hydrogen, the vector represents charge will be drawn from Hydrogen to Carbon. Similarly, as Nitrogen is more electronegative than Carbon, the vector will be towards Nitrogen from Carbon.
Despite quite a small difference in Carbon and Nitrogen’s electronegativities, it is considered a slightly polar bond as Nitrogen will try to pull the electrons to itself. Due to such differences, Hydrogen will have slightly positive charges, and Nitrogen will have slightly negative charges as the vector goes from Hydrogen to Nitrogen.
Thus Nitrogen becomes a negative pole, and the Hydrogen atom becomes a positive pole, making the molecular polar. Any molecule that has a difference of electronegativities of any dipole moment is considered as polar.
Hence, Hydrogen Cyanide is a polar molecule.
Concluding Remarks
To summarize everything in this article, we can say that:
- Carbon forms one single bond with the Hydrogen atom and forms a triple bond with the Nitrogen atom.
- HCN has a total of 10 valence electrons.
- It is covered under AX2 molecular geometry and has a linear shape.
- The bond angles of HCN is 180 degrees.
- Hydrogen Cyanide is a polar molecule.
Nitrogen is present in almost all proteins and plays important roles in both biochemical applications and industrial applications. Nitrogen forms strong bonds because of its ability to form a triple bond with its self, and other elements. Thus, there is a lot of energy in the compounds of nitrogen. Before 100 years ago, little was known about nitrogen. Now, nitrogen is commonly used to preserve food, and as a fertilizer.
Introduction
Nitrogen is found to have either 3 or 5 valence electrons and lies at the top of Group 15 on the periodic table. It can have either 3 or 5 valence electrons because it can bond in the outer 2p and 2s orbitals. Molecular nitrogen ((N_2)) is not reactive at standard temperature and pressure and is a colorless and odorless gas.
Nitrogen is a non-metal element that occurs most abundantly in the atmosphere, nitrogen gas (N2) comprises 78.1% of the volume of the Earth’s air. It only appears in 0.002% of the earth's crust by mass. Compounds of nitrogen are found in foods, explosives, poisons, and fertilizers. Nitrogen makes up DNA in the form of nitrogenous bases as well as in neurotransmitters. It is one of the largest industrial gases, and is produced commercially as a gas and a liquid.
| Name and Symbol | Nitrogen, N |
| Category | non-metal |
| Atomic Weight | 14.0067 |
| Group | 15 |
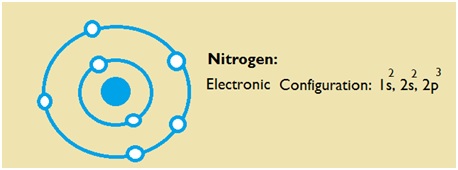
| Electron Configuration | 1s2 2s2 2p3 |
| Valence Electrons | 2, 5 |
| Phase | Gas |
History
Nitrogen, which makes up about 78% of our atmosphere, is a colorless, odorless, tasteless and chemically unreactive gas at room temperature. It is named from the Greek nitron + genes for soda forming. For many years during the 1500's and 1600's scientists hinted that there was another gas in the atmosphere besides carbon dioxide and oxygen. It was not until the 1700's that scientists could prove there was in fact another gas that took up mass in the atmosphere of the Earth.
Discovered in 1772 by Daniel Rutherford (and independently by others such as Priestly and Cavendish) who was able to remove oxygen and carbon dioxide from a contained tube full of air. He showed that there was residual gas that did not support combustion like oxygen or carbon dioxide. While his experiment was the one that proved that nitrogen existed, other experiments were also going in London where they called the substance 'burnt' or 'dephlogisticated air'.
Nitrogen is the fourth most abundant element in humans and it is more abundant in the known universe than carbon or silicon. Most commercially produced nitrogen gas is recovered from liquefied air. Of that amount, the majority is used to manufacture ammonia ((NH_3)) via the Haber process. Much is also converted to nitric acid ((HNO_3)).
Isotopes
Nitrogen has two naturally occurring isotopes, nitrogen-14 and nitrogen-15, which can be separated with chemical exchanges or thermal diffusion. Nitrogen also has isotopes with 12, 13, 16, 17 masses, but they are radioactive.
- Nitrogen 14 is the most abundant form of nitrogen and makes up more than 99% of all nitrogen found on Earth. It is a stable compound and is non-radioactive. Nitrogen-14 has the most practical uses, and is found in agricultural practices, food preservation, biochemicals, and biomedical research. Nitrogen-14 is found in abundance in the atmosphere and among many living organisms. It has 5 valence electrons and is not a good electrical conductor.
- Nitrogen-15 is the other stable form of nitrogen. It is often used in medical research and preservation. The element is non-radioactive and therefore can also be sometimes used in agricultural practices. Nitrogen-15 is also used in brain research, specifically nuclear magnetic resonance spectroscopy (NMR), because unlike nitrogen-14 (nuclear spin of 1), it has a nuclear spin of 1/2 which has benefits when it comes to observing MRI research and NMR observations. Lastly, nitrogen-15 can be used as label or in some proteins in biology. Scientists mainly use this compound for research purposes and have not yet seen its full potential for uses in brain research.
Compounds
The two most common compounds of nitrogen are Potassium Nitrate (KNO3) and Sodium Nitrate (NaNO3). These two compounds are formed by decomposing organic matter that has potassium or sodium present and are often found in fertilizers and byproducts of industrial waste. Most nitrogen compounds have a positive Gibbs free energy (i.e., reactions are not spontaneous).
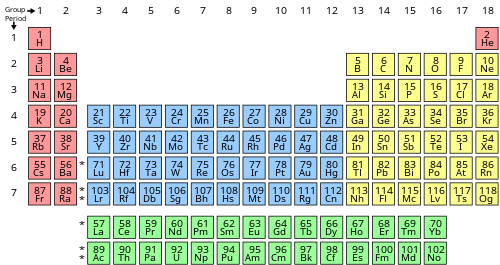
The dinitrogen molecule ((N_2)) is an 'unusually stable' compound, particularly because nitrogen forms a triple bond with itself. This triple bond is difficult hard to break. For dinitrogen to follow the octet rule, it must have a triple bond. Nitrogen has a total of 5 valence electrons, so doubling that, we would have a total of 10 valence electrons with two nitrogen atoms. The octet requires an atom to have 8 total electrons in order to have a full valence shell, therefore it needs to have a triple bond. The compound is also very inert, since it has a triple bond. Triple bonds are very hard to break, so they keep their full valence shell instead of reacting with other compounds or atoms. Think of it this way, each triple bond is like a rubber band, with three rubber bands, the nitrogen atoms are very attracted to each other.
Nitrides
Nitrides are compounds of nitrogen with a less electronegative atom; in other words it's a compound with atoms that have a less full valence shell. These compounds form with lithium and Group 2 metals. Nitrides usually has an oxidation state of -3.
[3Mg + N_2 rightarrow Mg_3N_2 label{1}]
When mixed with water, nitrogen will form ammonia and, this nitride ion acts as a very strong base.
[N + 3H_2O_{(l)} rightarrow NH_3 + 3OH^-_{(aq)} label{2}]
When nitrogen forms with other compounds it primarily forms covalent bonds. These are normally done with other metals and look like: MN, M3N, and M4N. These compounds are typically hard, inert, and have high melting points because nitrogen's ability to form triple covalent bonds.
Ammonium Ions
Nitrogen goes through fixation by reaction with hydrogen gas over a catalyst. This process is used to produce ammonia. As mentioned earlier, this process allows us to use nitrogen as a fertilizer because it breaks down the strong triple bond held by N2. The famous Haber-Bosch process for synthesis of ammonia looks like this:
[N_2 + 3H_2 rightarrow 2NH_3 label{3}]
Ammonia is a base and is also used in typical acid-base reactions.
[2NH_{3(aq)} + H_2SO_4 rightarrow (NH_4)_2SO_{4(aq)} label{4}]
Nitride ions are very strong bases, especially in aqueous solutions.
Oxides of Nitrogen
Nitrides use a variety of different oxidation numbers from +1 to +5 to for oxide compounds. Almost all the oxides that form are gasses, and exist at 25 degrees Celsius. Oxides of nitrogen are acidic and easily attach protons.
[N_2O_5 + H_2O rightarrow 2HNO_{3 (aq)} label{5}]
The oxides play a large role in living organisms. They can be useful, yet dangerous.
- Dinitrogen monoxide (N2O) is a anesthetic used at the dentist as a laughing gas.
- Nitrogen dioxide (NO2) is harmful. It binds to hemoglobin molecules not allowing the molecule to release oxygen throughout the body. It is released from cars and is very harmful.
- Nitrate (NO3-) is a polyatomic ion.
- The more unstable nitrogen oxides allow for space travel.
Hydrides
Hydrides of nitrogen include ammonia (NH3) and hyrdrazine (N2H4).
- In aqueous solution, ammonia forms the ammonium ion which we described above and it has special amphiprotic properties.
- Hyrdrazine is commonly used as rocket fuel
Applications of Nitrogen
- Nitrogen provides a blanketing for our atmosphere for the production of chemicals and electronic compartments.
- Nitrogen is used as fertilizer in agriculture to promote growth.
- Pressurized gas for oil.
- Refrigerant (such as freezing food fast)
- Explosives.
- Metals treatment/protectant via exposure to nitrogen instead of oxygen
References
- Petrucci, Ralph H, William Harwood, and F. Herring. General Chemistry: Principles and Modern Applications. 8th Ed. New Jersey: Pearson Education Inc, 2001.
- Sadava, David et al. LIFE: The Science of Biology. Eighth Edition. Sinauer Associate.
- Thomas, Jacob. Nitrogen and its Applications to Modern Future. San Diego State University Press: 2007.
Problems
- Complete and balance the following equations
N2+ ___H2→ ___NH_
H2N2O2 → ?
2NH3 + CO2 → ?
Nitrogen Electrons Valence Shell
__Mg + N2 → Mg_N_
N2H5 + H2O → ?
- What are the different isotopes of Nitrogen?
- List the oxiadation states of various nitrogen oxides: N2O, NO, N2O3, N2O4, N2O5
- List the different elements that Nitrogen will react with to make it basic or acidic....
- Uses of nitrogen
Answers
- Complete and balance the following equations
N2+ 3H2→ 2NH3(Haber process)
H2N2O2 → HNO
2NH3 + CO2 → (NH2)2CO + H2O
2Mg + 3N2 → Mg3N2
N2H5 + H2O → N2+ H+ + H2O

Nitrogen Valence Electrons Atom
- What are the different isotopes of Nitrogen?
Nitrogen Valence Electrons Count
Stable forms include nitrogen-14 and nitrogen-15
Nitrogen Valence Electrons Number
- List the oxidation states of various nitrogen oxides: +1, +2, +3, +4, +5 respectively
Nitrogen Valence Electrons
- List the different elements that Nitrogen will react with to make it basic or acidic :Nitride ion is a strong base when reacted with water, Ammonia is generally a weak acid
- Uses of nitrogen include anesthetic, Refrigerant, metal protector
